Abstract
Introduction
Acute promyelocytic leukemia (APL) is a very rare subtype of acute myeloid leukemia. APL might develop de novo or secondary after previous exposure to genotoxic agents or immunosuppression (sAPL). The data regarding sAPL is limited. The prognosis of sAPL patients (pts) and also the presence of any additional prognostic markers as compared to de novo APL, as well as the therapeutic approach still remain unclear. Here we present a case-matched analysis of sAPL diagnosed and treated according to the Polish Adult Leukemia Group/Programa Español de Tratamientos en Hematología (PALG/PETHEMA) protocols, compared with de novo APL.
Methods
The PALG's database of APL pts from 2006 to 2022 was screened and the cases of sAPL were selected. sAPL pts were matched based on sex, age, time of diagnosis, and treatment protocol with de novo APL pts from the same database. A variety of baseline variables as well as treatment outcomes were compared using U-Mann-Whitney, Chi2, Fisher tests, and Cox models.
Results
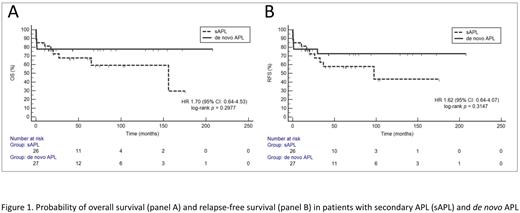
Out of 404 pts in the database, we found 27 pts with sAPL (6.7%), and 18 pts were female (66.6%). The median (Me) age in sAPL group was 58yo (range: 19-70yo). All reported cases of sAPL have developed after previous exposure to chemo- and/or radiotherapy. In 26 (96%) pts sAPL was proceeded with neoplasm treatment and in one (4%) multiple sclerosis therapy. The most often sAPL was diagnosed after treatment of breast cancer (29.6%), followed by therapy for kidney, colon, testis, lung, laryngeal, endometrial, ovarian cancers, and as well as Hodgkin lymphoma, B-acute lymphoblastic leukemia, liposarcoma, Merkel cell tumor, and GIST. In the analysis after pairs matching, we did not find any differences between sAPL and de novo APL regarding the ECOG performance status (p=0.29), WBC (Me 2.4G/L vs. 1.8G/L; p=0.52), coagulopathy described as a platelet counts (Me 24.5G/L vs. 37G/L, p=0.14), fibrinogen concentration (Me 1.0g/l vs. 1.4g/l, p=0.27) and d-dimers (Me 35.2ng/ml vs. 13.8ng/ml, p=0.23). We also did not observe any differences in additional cytogenetic abnormalities (p=0.13) between the examined groups. The frequency of FLT3-ITD expression in sAPL and de novo APL was 28.6% vs. 26.7%, respectively, (p=1.0), and FLT3-TKD expressions was 50% vs. 0%, respectively (p=0.43), however only a small number of pts had assessed FLT-TKD status (n=7 in both groups). The Me overall survival (OS) for the whole group was not reached. The Me OS for sAPL was 156 months (95%CI 26.9-156) and for de novo APL was not reached (p=0.29) (Figure 1A). In the univariate analysis, shorter OS was influenced by a high-risk group according to Sanz Score (HR 3.0, 95%CI 1.1-8; p<0.03), and ECOG 3 or 4 performance status at diagnosis (HR 11.52, 95%CI 3.8-34.9; p=<0.001). In the multivariate analysis shorter OS was associated with the presence of additional cytogenetic abnormalities other than t(15;17) (HR 16.8, 95%CI 2.7-103; p=0.002), older age (continuous variable) (HR 1.1, 95%CI 1-1.1; p=0.02) and lower platelets count (continuous variable) (HR 0.96, 95%CI 0.93-1.0; p=0.03). The relapse rate was not significantly higher in sAPL 19% (n=5) vs. de novo APL 3.7% (n=1)(p=0.2). The Me relapse-free survival (RFS) was not reached for the whole group. The Me RFS was 97.4 months (95%CI 26.3-97.4) in sAPL pts vs. not reached in de novo APL (p=0.31) (Figure 1B). Shorter RFS in the univariate analysis was affected by ECOG 3 or 4 at diagnosis and a high-risk group of Sanz Score (HR 8.6, 95%CI 3.1-23.8; p<0.001 and HR 4.3, 95%CI 1.6-10.8; p=0.002, respectively). In the multivariate analysis older age (continuous variable) (HR 1.1, 95%CI 1.0-1.1; p=0.03), lower platelets count (continuous variable) (HR 0.97, 95%CI 0.95-0.99; p=0.03) and additional cytogenetic abnormalities other than t(15;17) (HR 19.8, 95%CI 3.5-113.9; p=0.0008) negatively influenced RFS. sAPL diagnosis affected neither OS nor RFS. There are some limitations of our analysis, which include a low number of patients with sAPL and a lack of prospective data.
Conclusions
The results of this case-matched study are in line with the previous suggestions that outcomes of pts with sAPL may be similar to those with the disease de novo. We also conclude that there is no difference in the clinical and biological presentation of sAPL and de novo APL at the time of diagnosis.
Disclosures
Pluta:Angelini: Honoraria; Astellas: Honoraria; Celgen/BMS: Honoraria; Novartis: Honoraria; Swixx Biopharma: Honoraria, Research Funding. Sobas:Celgene/BMS: Honoraria; Novartis: Honoraria. Krawiec:Celgene/BMS: Honoraria. Czemerska:Novartis: Honoraria; Pfizer: Honoraria; Celgene: Honoraria; Sandoz: Honoraria; Abbvie: Honoraria. Wróbel:GSK: Honoraria; Abbvie: Honoraria; Gilead: Honoraria; Celgen/BMS: Honoraria; Roche: Honoraria, Research Funding; Novartis: Honoraria; Janssen: Honoraria; Amgen: Honoraria, Research Funding; Takeda: Honoraria; Beigene: Honoraria. Watek:Novartis: Honoraria. Zaucha:Takeda: Honoraria; Gilead: Honoraria; Novartis: Honoraria; BMS: Research Funding; Abbvie: Honoraria; Roche: Honoraria; Janssen: Honoraria; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Consultancy. Helbig:Novartis: Honoraria. Oleksiuk:Novo Nordisk: Speakers Bureau; Roche: Consultancy, Speakers Bureau; Takeda: Speakers Bureau. Gil:Janssen: Honoraria; Astellas: Honoraria; Gilead: Honoraria; Abbvie: Honoraria; Celgene/BMS: Honoraria; Novartis: Honoraria; Pfizer: Honoraria. Wierzbowska:Janssen: Honoraria; Gilead: Honoraria; AbbVie: Honoraria; Celgene/BMS: Honoraria; Astellas: Honoraria; Novartis: Honoraria; Swixx Biopharma: Honoraria, Research Funding; Servier: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal